Adapted from cushings-help.com and The Merck Manual
Adrenal Gland Disorders
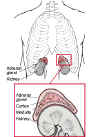 The body has two adrenal glands, one near the top of each kidney. The inner part (medulla) of the adrenal glands secretes hormones such as adrenaline (epinephrine) that affect blood pressure, heart rate, sweating, and other activities also regulated by the sympathetic nervous system. The outer part (cortex) secretes many different hormones, including corticosteroids (cortisone-like hormones), androgens (male hormones), and mineralocorticoids, which control blood pressure and the levels of salt and potassium in the body.
The body has two adrenal glands, one near the top of each kidney. The inner part (medulla) of the adrenal glands secretes hormones such as adrenaline (epinephrine) that affect blood pressure, heart rate, sweating, and other activities also regulated by the sympathetic nervous system. The outer part (cortex) secretes many different hormones, including corticosteroids (cortisone-like hormones), androgens (male hormones), and mineralocorticoids, which control blood pressure and the levels of salt and potassium in the body.
The adrenal glands are part of a complex system that produces interacting hormones. The hypothalamus produces corticotropin-releasing hormone, triggering the pituitary gland to secrete corticotropin, which regulates the production of corticosteroids by the adrenal glands. Adrenal glands may stop functioning when either the pituitary or hypothalamus fails to produce sufficient amounts of the appropriate hormones. Underproduction or overproduction of any adrenal hormones can lead to serious illness.
Underactive Adrenal Glands
Addison's disease (adrenocortical insufficiency) results when underactive adrenal glands produce insufficient amounts of corticosteroids.
Addison's disease affects about 4 out of every 100,000 people. The disease can strike at any age and affects males and females about equally. In 30 percent of people with Addison's disease, the adrenal glands are destroyed by a cancer, amyloidosis, an infection such as tuberculosis, or another identifiable disease. In the other 70 percent, the cause isn't known for certain, but scientists strongly suspect the adrenal glands are destroyed by an autoimmune reaction.
The adrenal glands are also suppressed in people who take corticosteroids such as prednisone. Ordinarily, the dose of corticosteroids is tapered slowly before the drug is stopped completely. When corticosteroids are stopped suddenly after being taken for a month or more, the adrenal glands may be unable to produce corticosteroids in sufficient amounts for several weeks or even months, depending on the dose of corticosteroids and the duration of treatment. Certain other drugs, such as ketoconazole taken to treat fungal infections, can also block the natural production of corticosteroids, resulting in a deficiency.
Corticosteroid deficiency can lead to many problems. For example, when corticosteroids are lacking, the body excretes large amounts of sodium and retains potassium, leading to low levels of sodium and high levels of potassium in the blood. The kidneys aren't able to concentrate urine, so when a person with a corticosteroid deficiency drinks too much water or loses too much sodium, the blood level of sodium falls. Inability to concentrate urine ultimately causes the person to urinate excessively and become dehydrated. Severe dehydration and a low sodium level reduce blood volume and can culminate in shock.
corticosteroid deficiency also leads to an extreme sensitivity to insulin, a hormone normally present in the blood, so that the blood sugar levels may fall dangerously low. The deficiency prevents the body from manufacturing carbohydrates from protein, fighting infections, or healing wounds very well. Muscles weaken, and even the heart can become weak and unable to pump blood adequately.
To compensate for a deficiency of corticosteroids, the pituitary gland produces more corticotropin, the hormone that normally stimulates the adrenal glands. Since corticotropin also affects melanin production, people with Addison's disease often develop a dark pigmentation of the skin and the lining of the mouth. The excessive pigmentation usually occurs in patches. Even people with dark skin can develop excessive pigmentation, although the change may be hard to recognize. Excessive pigmentation doesn't occur when adrenal insufficiency is caused by pituitary or hypothalamus insufficiency, conditions in which the basic problem is a deficiency of corticotropin.
Symptoms
Soon after developing Addison's disease, a person feels weak, tired, and dizzy when standing up after sitting or lying down. The skin becomes dark; this darkness may seem like tanning, but it appears on both sun-exposed and nonexposed areas. Black freckles may develop over the forehead, face, and shoulders; a bluish-black discoloration may develop around the nipples, lips, mouth, rectum, scrotum, or vagina. Most people lose weight, become dehydrated, have no appetite, and develop muscle aches, nausea, vomiting, and diarrhea. Many become unable to tolerate cold. Unless the disease is severe, symptoms tend to become apparent only during times of stress.
If the disease isn't treated, severe abdominal pains, profound weakness, extremely low blood pressure, kidney failure, and shock may occur, especially if the body is subjected to stress such as an injury, surgery, or severe infection. Death may quickly follow.
Diagnosis
Because the symptoms may start slowly and be subtle, and because no single laboratory test is definitive, doctors often don't suspect Addison's disease at the outset. Sometimes a major stress, such as an accident, operation, or serious illness, makes the symptoms more obvious and precipitates a crisis.
Blood tests may show a lack of corticosteroids, especially cortisol, as well as low sodium and high potassium levels. Measures of kidney function, such as tests for blood urea nitrogen and creatinine, usually indicate that the kidneys aren't working well. Corticosteroid levels, usually tested after an injection of corticotropin (a challenge test), can help the doctor distinguish adrenal gland insufficiency from pituitary gland insufficiency. When it is the latter, an injection of corticotropin-releasing hormone reveals whether the cause of the problem is hypothalamus insufficiency.
Treatment
Regardless of the cause, Addison's disease can be life-threatening and must be treated first with corticosteroids. Usually treatment can be started with prednisone taken orally. However, people who are severely ill may be given cortisol intravenously at first and then prednisone tablets. Most people with Addison's disease also need to take 1 or 2 tablets of fludrocortisone every day to help restore the body's normal excretion of sodium and potassium. Fludrocortisone can eventually be reduced or discontinued in some people; however, they will need to take prednisone every day for the rest of their lives. Larger doses of prednisone may be needed when the body is stressed, especially from an illness. Although treatment must be continued for life, the outlook for a normal life span is excellent.
Overactive Adrenal Glands
The adrenal glands can produce too much of one or more hormones. Changes in the adrenal glands themselves or overstimulation by the pituitary gland may be the cause. The symptoms and treatment depend on which hormones--androgenic steroids, corticosteroids, or aldosterone--are being overproduced.
Overproduction of Androgenic Steroids
Overproduction of androgenic steroids (testosterone and similar hormones) is a condition that leads to virilization, the development of exaggerated masculine characteristics in either men or women.
Mild overproduction of androgens is common but may lead only to increased hair growth (hirsutism). True virilizing disease is rare, affecting only about 1 or 2 of every 100,000 women. The incidence of virilizing disease in men is almost impossible to guess.
Symptoms
Signs of virilization include hairiness of the face and body, baldness, acne, deepening of the voice, and increased muscularity. In women, the uterus shrinks, the clitoris enlarges, the breasts become smaller, and normal menstruation stops. Both men and women may experience an increased sex drive.
Diagnosis
The combination of body changes makes virilization relatively easy for a doctor to recognize. A test can determine the level of androgenic steroids in the urine. If the level is high, the dexamethasone suppression test can help determine whether the problem is a cancer, a noncancerous tumor (adenoma), or an enlargement of the hormone-producing portions of the adrenal cortex (adrenal hyperplasia). With this test, the corticosteroid dexamethasone is given orally. If the problem is adrenal hyperplasia, dexamethasone prevents the adrenal glands from producing androgenic steroids. If the problem is an adenoma or cancer of the adrenal glands, dexamethasone reduces androgenic steroid production only partially or not at all. The doctor may also order a computed tomography (CT) or magnetic resonance imaging (MRI) scan to obtain a view of the adrenal glands.
Treatment
Androgen-producing adenomas and adrenal cancers are usually treated by surgically removing the adrenal gland. For adrenal hyperplasia, small amounts of corticosteroids such as dexamethasone generally reduce the production of androgenic steroids, but these drugs may also cause symptoms of Cushing's syndrome if too large a dose is given.
Overproduction of Corticosteroids
Overexposure to corticosteroids, whether from overproduction by the adrenal glands or from administration of excessive amounts by a doctor, results in Cushing's syndrome.
An abnormality in the pituitary gland, such as a tumor, can cause the pituitary to produce large amounts of corticotropin, the hormone that controls the adrenal glands. Pituitary tumors that overproduce corticotropin occur in about 6 in every 1 million people. Small-cell carcinoma in the lung and some other tumors outside the pituitary gland can produce corticotropin as well (a condition called ectopic corticotropin syndrome). This is the most common cause of excessive adrenal cortical function, found in at least 10 percent of people with small-cell carcinoma in the lung, a common type of tumor.
Sometimes the adrenal gland produces excessive corticosteroids even when corticotropin levels are low, usually when a benign tumor (adenoma) has developed in the adrenal gland. Benign tumors of the adrenal cortex are extremely common; half of all people have them by the age of 70. Only a small fraction of these benign tumors are active; the incidence of adenomas causing disease is about 2 in every 1 million people. Cancerous tumors of the adrenal cortex are equally common, but cancers causing endocrine disease are quite rare.
Symptoms
Because corticosteroids alter the amount and distribution of body fat, a person with Cushing's syndrome usually has a large, round face (moon face). Excessive fat develops throughout the torso and may be particularly noticeable at the top of the back (buffalo hump). Fingers, hands, and feet are usually slender in proportion to the thickened trunk. Muscles lose their bulk, leading to weakness. The skin becomes thin, bruises easily, and heals poorly when bruised or cut. Purple streaks that look like stretch marks may develop over the abdomen.
High corticosteroid levels over time raise the blood pressure, weaken bones (osteoporosis), and diminish resistance to infections. The risk of developing kidney stones and diabetes is increased, and mental disturbances, including depression and hallucinations, may occur. Women with Cushing's syndrome usually have an irregular menstrual cycle. Children who have the condition grow slowly and remain short. In some people, the adrenal glands also produce large amounts of androgenic steroids, leading to increased facial and body hair, balding, and an increased sex drive.
Diagnosis
Doctors who suspect Cushing's syndrome after observing the symptoms measure the blood level of cortisol, the main corticosteroid hormone. Normally, cortisol levels are high in the morning and decrease during the day. In people who have Cushing's syndrome, cortisol levels are very high in the morning and don't decrease late in the day as would be expected. Measuring cortisol in the urine can be useful because tests performed a few hours apart can indicate how much cortisol has been produced in that time.
If the cortisol levels are high, the doctor may recommend a dexamethasone suppression test. The test is based on the ability of dexamethasone to suppress the pituitary gland, thereby reducing adrenal gland stimulation. First a urine sample is tested for cortisol. Then dexamethasone is given, and cortisol levels are measured in another sample of urine. If the Cushing's syndrome is caused by pituitary stimulation, the level of cortisol will fall; if the Cushing's syndrome is caused by stimulation from a nonpituitary source of corticotropin or an adrenal tumor, the urinary cortisol level will remain high.
Results of a dexamethasone suppression test may not be clear-cut. Other laboratory tests may be needed to help determine the precise cause of the syndrome. These tests may be followed by a computed tomography (CT) or magnetic resonance imaging (MRI) scan of the pituitary or adrenal glands and by a chest x-ray or CT scan of the lungs.
Treatment
Treatment is directed at the pituitary or adrenal gland depending on the source of the problem. Surgery or radiation therapy may be needed to remove or destroy a pituitary tumor. Adenomas of the adrenal gland can often be removed surgically. Both adrenal glands may have to be removed if these treatments aren't effective or if no tumor is present. Any person who has had both adrenal glands removed, and many people who have had part of their adrenal glands removed, must take corticosteroids for life.
Some 5 to 10 percent of the people who have both adrenal adrenal glands removed develop Nelson's syndrome. In this condition, the pituitary gland enlarges, producing large amounts of corticotropin and other hormones such as beta-melanocyte-stimulating hormone, which darkens the skin. If necessary, Nelson's syndrome can be treated with radiation or surgical removal of the pituitary gland.
Overproduction of Aldosterone
Overproduction of aldosterone (hyperaldosteronism) by the adrenal glands is a condition that affects the blood levels of sodium, potassium, bicarbonate, and chloride, leading to high blood pressure, weakness, and, rarely, periods of paralysis.
Aldosterone, a hormone produced and secreted by the adrenal glands, signals the kidney to excrete less sodium and more potassium. Aldosterone production is regulated partly by corticotropin in the pituitary and partly by a control mechanism in the kidneys (the renin-angiotensin-aldosterone system). Renin, an enzyme produced in the kidneys, controls the activation of the hormone angiotensin, which stimulates the adrenal glands to produce aldosterone.
Hyperaldosteronism can be caused by a tumor (usually noncancerous) in the adrenal gland (a condition called Conn's syndrome). Sometimes hyperaldosteronism is a response to certain diseases. For example, the adrenal glands secrete large amounts of aldosterone if the blood pressure is very high or if the artery that carries blood to the kidneys is narrowed.
Symptoms
High levels of aldosterone can lead to low levels of potassium, causing weakness, tingling, muscle spasms, and paralysis. The nervous system may not function properly. Some people become extremely thirsty and urinate frequently, and some experience personality changes.
Symptoms of hyperaldosteronism are also associated with eating licorice, which contains a chemical very similar to aldosterone. In rare cases, people who eat a great deal of candy with real licorice flavoring may develop all the symptoms of hyperaldosteronism.
Diagnosis and Treatment
A doctor who suspects that high blood pressure or related symptoms are caused by hyperaldosteronism may measure the sodium and potassium levels in the blood. The doctor may also measure aldosterone levels and, if they're high, may prescribe spironolactone, a drug that blocks the action of aldosterone, to see if the levels return to normal. Other tests generally aren't needed.
When too much aldosterone is being produced, doctors examine the adrenal glands for an adenoma or cancer. While a computed tomography (CT) or magnetic resonance imaging (MRI) scan can be helpful, exploratory surgery is often necessary. If a growth is found, it can usually be removed. When a simple adenoma is removed, blood pressure returns to normal and other symptoms disappear about 70 percent of the time. If no tumor is found and the entire gland is overactive, partial removal of the adrenal glands may not control high blood pressure and complete removal will produce adrenal insufficiency, requiring treatment for the rest of the person's life. However, spironolactone can usually control the symptoms, and drugs for high blood pressure are readily available. Rarely do both adrenal glands have to be removed.
More:
Addison’s Disease Knol at http://knol.google.com/k/addison-s-disease#
Addison's Disease (Adrenal insufficiency): Caused by low levels of cortisonelike hormones produced by the adrenal glands. The cause is usually unknown, but may be a complications of tuberculosis, cancer, pituitary disease of cortisone drugs.
Addison's Disease
 This Topic on the Message Boards.
This Topic on the Message Boards.
Adrenal: Pertaining to one or both of these endocrine glands located on top of the kidneys. These glands secrete many hormones, including epinephrine (adrenaline), norepinephrine, and the corticosteroid, and play an important part in the body's endocrine system.
The adrenal is made up of an outer wall (the cortex) and an inner portion (the medulla).
A closer look at the adrenal glands, including an illustration.
Adrenal cortex: Outer layer of the adrenal gland, it secretes various hormones including cortisone, estrogen, testosterone, cortisol, androgen, aldosterone and progesterone.
Adrenalectomy: Surgical removal of the adrenal glands is a final measure for halting excess cortisol production This is used only when all other measures fail in individuals with pituitary tumors.
Adrenal gland tumor: A benign tumor, or adenoma, that usually results in the excess production of adrenal gland hormones.
Sometimes, an abnormality of the adrenal glands, most often an adrenal tumor, causes Cushing's syndrome. The average age of onset is about 40 years. Most of these cases involve non-cancerous tumors of adrenal tissue, called adrenal adenomas, which release excess cortisol into the blood. adrenal glands. In Primary Pigmented Micronodular Adrenal Disease and the familial Carney's complex, surgical removal of the adrenal glands is required.
Adrenal insufficiency: See Addison's Disease. Adrenal insufficiency is a life threatening chronic illness. An active and vigorous lifestyle with normal life expectancy is possible as long as the prescribed medications are taken regularly and adjusted when indicated. As with most chronic diseases, adrenal insufficiency demands that the patients take responsibility and develop self-management skills and techniques.
Read an article on Adrenal insufficiency
Adrenal medulla: Middle part of the adrenal gland, it secretes epinephrine (adrenalin) and norepinephrine.
Adrenocortical carcinomas, or adrenal cancers: These are the least common cause of Cushing's syndrome. Cancer cells secrete excess levels of several adrenal cortical hormones, including cortisol and adrenal androgens. Adrenocortical carcinomas usually cause very high hormone levels and rapid development of symptoms.
Adrenocortical hyperplasia: Increase in the number of cells of the adrenal cortex. It secretes cortisol, androgens and aldosterone. Increased production of any or all of these hormones may result in a variety of disorders, such as Cushing's syndrome and hypertension.
Adrenocorticotropic-hormone deficiency: Not enough ACTH is produced by the pituitary gland.
Adrenoleukodystrophy: Disturbance in brain substance caused by abnormal function of the adrenal gland.
Aldosterone: Adrenal hormone that affects the body's handling of sodium, chloride, and potassium.
Read an article on Aldosterone testing












