Labels: Cushings Help, forums, message boards, upgrade
David Unuane, MD (Doctor), Department of Endocrinology, Universitair Ziekenhuis Brussel, UZ Brussel, Vrije Universiteit Brussel, Brussels, Belgium, Herman Tournaye, MD, PhD (Professor, Doctor), Brigitte Velkeniers, MD, PhD (Professor, Doctor), Kris Poppe, MD, PhD (Professor, Doctor)
Female infertility occurs in about 37% of all infertile couples and ovulatory disorders account for more than half of these. The ovaries are in continuous interaction with the other endocrine organs. The interplay may account for infertility occurring at different levels and may render the diagnosis of infertility a difficult exercise for the involved physician. A hypothalamic cause of female infertility should be considered in an appropriate clinical context, with tests pointing to a hypogonadotropic hypogonadism. It can be functional, physiological or related to organic causes. Hyperprolactinemia has well characterized effects on the normal gonadal function and treatment is well established.
Acromegaly and Cushing’s disease may impair fertility at different levels, mechanisms involved however remain ill defined. Thyroid disorders, both hyperthyroidism and hypothyroidism, can interact with the ovaries, through a direct effect on ovarian function, but autoimmunity may be involved, as well as alterations of the sex hormone binding protein levels. Primary ovarian disorders, such as the polycystic ovary syndrome and primary ovarian insufficiency are frequent diseases, for which novel treatments are currently being developed and discussed.
We will propose an algorithm for the diagnosis and approach of the female patient presenting with infertility on the basis of the available evidence in literature.
Keywords: female infertility, pituitary, adrenal, ovarian, thyroid
From http://www.bprcem.com/article/PIIS1521690X1100087X/abstract?rss=yes
Labels: acromegaly, adrenal, Cushing's, hyperprolactinemia, infertility, PCOS, pituitary, thyroid
Source: Johns Hopkins Medicine
FOR IMMEDIATE RELEASE
Newswise — Infertility is common among obese women, but the reasons remain poorly understood and few treatments exist. Now a team of Johns Hopkins Children's Center scientists, conducting experiments in mice, has uncovered what they consider surprising evidence that insulin resistance, long considered a prime suspect, has little to do with infertility in women with type-2 diabetes, polycystic ovary syndrome (PCOS) and metabolic syndrome, all obesity-related conditions in which the body becomes desensitized to insulin and loses the ability to regulate blood sugar.
In a report, published online Nov.10 in the journal Diabetes, the Johns Hopkins scientists say the real culprit appears to be insulin sensitivity in the ovaries and the pituitary.
The Johns Hopkins team said its findings show that these organs escape insulin resistance and, awash with high levels of circulating insulin common in obesity, develop abnormal cell signaling that disrupts ovulation and eventually leads to infertility.
"Our findings suggest that the focus should shift from treating insulin resistance in peripheral tissue to taming insulin sensitivity in the pituitary and ovaries," says lead investigator Sheng Wu, Ph.D., of the Johns Hopkins Children's Center. Scientists traditionally have treated obesity-induced infertility by lowering blood insulin to counter the effects of insulin resistance.
A 2010 study by the same team discovered that the pituitary gland, insensitive to insulin in lean mice, became sensitive to elevated levels of insulin seen in human and rodent obesity. By knocking out the insulin receptors in the pituitary glands of obese mice, the researchers were able to partially restore fertility, thus proving that abnormal insulin signaling in the pituitary was only part of the story.
"In the original study, disrupting insulin signaling in the pituitary restored 50 percent of fertility in obese mice, but the search was on for the accomplice," says senior investigator Andrew Wolfe, Ph.D., an endocrinologist at the Johns Hopkins Children's Center. "Our new findings point to the ovaries."
In the pituitary, faulty insulin signaling stimulates increased secretion of luteinizing hormone, the researchers say. In the ovary, it puts testosterone production into overdrive. Both disrupt ovulation, the researchers explain.
In the latest study, lean mice and mice made obese on a three-month high-fat diet received injections of progressively higher doses of insulin to mimic the effects of high circulating insulin seen in obesity, diabetes and PCOS. In lean mice, the ovaries and pituitaries were insensitive to the hormone at low-dose injections, and responded only when injected with higher doses of insulin. The "trigger" doses corresponded to insulin levels typically seen in obesity. Obese mice with naturally elevated insulin levels exhibited high levels of insulin signaling in their pituitary and ovarian cells. When injected with insulin, the livers and muscles of obese mice showed greatly reduced response to insulin -- or insulin resistance. Their ovaries and pituitary glands, however, responded to insulin injections, confirming that in obese mice, these reproductive organs escape the insulin resistance seen in other organs.
To determine insulin sensitivity, the researchers focused on two signaling proteins, IRS-1 and IRS-2, regulators of cell-insulin communication involved in the development of insulin resistance in liver and muscle tissue. The scientists hypothesized that in the pituitary and ovaries, these messenger proteins would remain dormant under normal insulin levels, but would get activated once exposed to high levels of insulin. Indeed, the researchers found, the pituitary glands of obese mice showed higher IRS-2 signaling activity compared with lean mice, while the ovaries of obese mice had higher signaling activity in both IRS-1 and IRS-2 proteins, compared with lean mice.
In a follow-up study now under way, the Hopkins team is trying to determine whether knocking out the insulin receptors in both the ovaries and the pituitary would fully restore fertility in obese mice with high insulin levels.
Other co-investigators on the study included Sara Divall, M.D., and Fred Wondisford, M.D., both of the Johns Hopkins Children's Center.
The research was funded by the Endocrine Fellow Foundation, by The Eunice Kennedy Shriver National Institute of Child Health and Human Development, part of the National Institutes of Health, and by the Baltimore Diabetes Research and Training Center, which is supported by the National Institute for Diabetes and Digestive and Kidney Diseases.
Founded in 1912 as the children's hospital of the Johns Hopkins Medical Institutions, the Johns Hopkins Children's Center offers one of the most comprehensive pediatric medical programs in the country, treating more than 90,000 children each year. Hopkins Children's is consistently ranked among the top children's hospitals in the nation.
Hopkins Children's is Maryland's largest children's hospital and the only state-designated Trauma Service and Burn Unit for pediatric patients. It has recognized Centers of Excellence in dozens of pediatric subspecialties, including allergy, cardiology, cystic fibrosis, gastroenterology, nephrology, neurology, neurosurgery, oncology, pulmonary, and transplant. For more information, please visitwww.hopkinschildrens.org
From http://www.newswise.com/articles/not-the-usual-suspects-animal-study-finds-su.../articles/list&category=medicine&page=1&search%5Bstatus%5D=3&search%5Bsort%5D=date+desc&search%5Bsection%5D=10&search%5Bhas_multimedia%5D=
By Lisa Smyth
A Northern Ireland man has defied medical predictions and overcome the effects of a rare condition that left him in a coma for three months.
Jonathan Fisher is a survivor of an Addisonian crisis which affected him so badly doctors feared he would never recover.
His mother ignored medical advice to switch off his life-support system as doctors believed he was brain dead.
However, convinced he could recover after noticing that he was reacting to her with the occasional slight movement of his little finger she refused to give up on her son.
Now — against all the odds — he has made a remarkable recovery and has regained many of the skills lost as a result of his condition.
Addison’s disease is a rare disorder of the adrenal glands. It affects the production of two hormones — cortisol and aldosterone — which help to regulate blood pressure.
If left untreated, the amount of steroid hormones in the body will gradually fall and the symptoms of Addison’s disease will get progressively worse.
Eventually, this will cause an adrenal crisis — when the symptoms become very severe and blood pressure drops to a dangerously low level. An adrenal crisis can be fatal if it is not treated immediately.
Jonathan can now get around Lisburn in his electric wheelchair and has even managed to complete his first book, August Always — a triumph as he has considerable speech impairment and great difficulty using his hands to operate a keyboard.
The process has been long and laborious but Jonathan said he was determined to share his experience with others.
“There are moments in life that define us, like birth and death,” he said.
“Along the journey there will be crises of passion, of love, of faith and desire, but none so devastating as an Addisonian crisis. I am Jonathan Fisher, a survivor.
“August Always is my memoir. I believe in the incredible. I dream of a better future.”
Background
Addison’s disease is a rare disorder of the adrenal glands which are located on top of the kidneys. The condition affects the production of two hormones. Cortisol, which helps to regulate blood pressure, maintaining blood glucose and heart function. And aldosterone which also helps regulate blood pressure. Addison’s sufferers must get treatment if their blood pressure falls as it can be fatal.
From http://www.belfasttelegraph.co.uk/news/health/coma-man-defies-odds-to-write-book-16076458.html
Labels: Addison's, adrenal, Adrenal Crisis, steroids
Diabetes mellitus is a frequent complication of Cushing syndrome (CS) which is caused by chronic exposure to glucocorticoid excess, either endogenous or exogenous, and that is characterized by several clinical symptoms such as central obesity, purple striae, proximal muscle weakness, acne, hirsutism and neuropsychological disturbances.
Diabetes occurs as a consequence of an insulin-resistant state together with impaired insulin secretion which are induced by glucocorticoid excess. The management of patients with CS and diabetes mellitus includes the treatment of hyperglycemia and, when possible, the correction of glucocorticoid excess.
This review focuses on the disorders of glucose metabolism in patients exposed to glucocorticoid excess, addressing both the pathophysiological aspects and the clinical and therapeutic implications.
Read the entire article at http://www.cell.com/trends/endocrinology-metabolism/abstract/S1043-2760(11)00138-X
Labels: acne, Cushing's, diabetes, diabetes mellitus, glucocorticoids, hirsuitism, insulin, obesity, straie
Something new of interest to Cushies most every day. Please note that there is a current backlog of about three weeks for submitted bios to be added to the website.
November 10, 2011:
November 5, 2011:
November 2, 2011:
Labels: adrenal, bios, CushieWiki, Cushing's, FAQ, news items, pituitary
HELSINGBORG, Sweden and EXTON, Pa., Nov. 7, 2011 /PRNewswire/ -- The Swedish specialty pharma company, DuoCort Pharma, announced today that the European Commission has granted a European Marketing Authorisation for Plenadren®(hydrocortisone, modified release tablet), an orphan drug for treatment of adrenal insufficiency in adults, bringing these patients their first pharmaceutical innovation in over 50 years.
Developed by DuoCort Pharma, Plenadren® is a dual release hydrocortisone replacement therapy designed to better mimic the normal physiological cortisol profile in order to improve outcomes for patients suffering from adrenal insufficiency. Plenadren® is given as an oral tablet once daily. It has an outer layer releasing hydrocortisone immediately and an inner core releasing the rest of the drug more slowly during the day.
Although glucocorticoid hormone replacement therapy for adrenal insufficiency has been available for decades, studies have recorded complications and comorbidities including premature death, impaired quality of life, increased risk of cardiovascular diseases, and decreased bone mineral density in treated patients, most likely because it is difficult to match the natural secretion pattern of cortisol.
Maria Forss, CEO of DuoCort Pharma, said: "The marketing authorization for Plenadren® in Europe is an important step towards addressing the unmet needs of these patients."
The approval of Plenadren® follows the positive opinion adopted by the Committee for Medicinal Products for Human Use (CHMP) in July 2011. Plenadren® is now approved for marketing in all countries of the European Union (EU) as well as in the European Economic Area (EEA), namely Iceland, Norway and Lichtenstein.
Professor Gudmundur Johannsson of the Department of Endocrinology, Sahlgrenska University Hospital, Gothenburg, Sweden, and Chief Medical Officer of DuoCort Pharma, said: "Plenadren® offers a welcome new treatment option to help patients suffering from adrenal insufficiency. Plenadren® can improve therapy for many of the almost 200,000 patients in Europe who suffer from this disease and who need life-long cortisol replacement therapy for their survival."
On October 26, 2011, ViroPharma Incorporated (NASDAQ: VPHM) signed a definitive agreement to acquire DuoCort Pharma AB. The companies expect to complete the acquisition in November 2011. On closing, ViroPharma will pay an upfront closing cost of 220 million Swedish kroner (SEK) or $33 million in US dollars (USD). Additionally, there are contingent milestone payments of up to 860 million SEK or $130 million USD associated with manufacturing, sales thresholds and territory expansion.
About Adrenal insufficiency
Adrenal insufficiency (cortisol deficiency) is a rare, life-threatening disease that affects patients in their active years. To survive, patients suffering from this disease need lifelong replacement therapy with hydrocortisone. Treatment of adrenal insufficiency involves replacing, or substituting, the hormones that the patient's own adrenal glands are not producing. Cortisol is replaced using hydrocortisone, the synthetic form of cortisol.
About Plenadren® (hydrocortisone, modified release tablet)
Plenadren is the first true innovation in over 50 years in the treatment of adrenal insufficiency.
Hypersensitivity to the active substance of Plenadren or to any of the excipients may occur. During acute adrenal insufficiency, parenteral administration of hydrocortisone in high doses, together with physiological sodium chloride solution for injection, must be given. Use of Plenadren with potent CYP 3A4 inducers and inhibitors may merit an adjustment of hydrocortisone dosage. High (supra-physiological) dosages of cortisone can cause elevation of blood pressure, salt and water retention, and increased excretion of potassium. Long-term treatment with higher than physiological hydrocortisone doses can lead to clinical features resembling Cushing's syndrome with increased adiposity, abdominal obesity, hypertension and diabetes, and thus result in an increased risk of cardiovascular morbidity and mortality. All glucocorticoids increase calcium excretion and reduce the bone remodeling rate. Patients with adrenal insufficiency on long term glucocorticoid replacement therapy have been found to have reduced bone mineral density. Psychiatric adverse events may occur with systemic glucocorticoids.
The most common adverse reactions observed in clinical studies have been fatigue, gastroenteritis, upper respiratory tract infection, sedation, vertigo and dry eyes.
About ViroPharma Incorporated
ViroPharma Incorporated is an international biopharmaceutical company committed to developing and commercializing novel solutions for physician specialists to address unmet medical needs of patients living with diseases that have few if any clinical therapeutic options, including C1 esterase inhibitor deficiency, treatment of seizures in children and adolescents, and C. difficile infection (CDI). Our goal is to provide rewarding careers to employees, to create new standards of care in the way serious diseases are treated, and to build international partnerships with the patients, advocates, and health care professionals we serve. ViroPharma's commercial products address diseases including hereditary angioedema (HAE), seizures in children and adolescents, and CDI; for full U.S. prescribing information on our products, please download the package inserts athttp://www.viropharma.com/Products.aspx; the prescribing information for other countries can be found atwww.viropharma.com.
ViroPharma routinely posts information, including press releases, which may be important to investors in the investor relations and media sections of our company's web site, www.viropharma.com. The company encourages investors to consult these sections for more information on ViroPharma and our business.
About DuoCort Pharma
DuoCort Pharma is a drug development company focused on improving glucocorticoid therapy. The company has its origins among researchers at the Sahlgrenska Academy at Gothenburg University and at Uppsala University in Sweden. DuoCort Pharma has developed Plenadren®, an improved glucocorticoid replacement therapy for patients with adrenal insufficiency, which is a rare disease. DuoCort Pharma has orphan drug designations in EU, Switzerland and the USA for Plenadren®. Plenadren® is a once daily, dual-release hydrocortisone oral tablet. It has an outer layer that releases the drug immediately and an inner core that releases the drug over the day. The tablets come in both 5 mg and 20 mg strengths. For more information please visit www.duocort.com.
DuoCort Pharma is a project company of the life science incubator PULS. For more information visit www.pulsinvest.se.
Disclosure Notice
Certain statements in this press release contain forward-looking statements that involve a number of risks and uncertainties. Forward-looking statements provide our current expectations or forecasts of future events, including statements about the benefits of the business combination transaction involving ViroPharma and DuoCort Pharma, including, among others, future financial and operating results, enhanced revenues, ViroPharma's plans, objectives, expectations and intentions and other statements that are not historical facts. The following factors, among others, could cause actual results to differ from those set forth in the forward-looking statements: the ability to achieve the other conditions to closing on the proposed schedule; the risk that the business will not be integrated successfully; the risk that revenues following the acquisition will be lower than expected, including the successful commercialization of Plenadren; potential for disruption from the transaction making it more difficult to maintain relationships with manufacturers, employees or other suppliers; competition and its effect on pricing, spending, third-party relationships and revenues; our ability to achieve favorable pricing for Plenadren from European regulatory authorities; the risk that the safety and/or efficacy results of existing clinical trials for Plenadren will not be consistent with the results of additional clinical studies, including the required registry study, or with commercial usage; market acceptance of Plenadren; and our inability to maintain the orphan drug status associated with Plenadren. These factors, and other factors, including, but not limited to those described in our annual report on Form 10-K for the year ended December 31, 2010 and quarterly reports on Form 10-Q filed with the Securities and Exchange Commission, could cause future results to differ materially from the expectations expressed in this press release. The forward-looking statements contained in this press release are made as of the date hereof and may become outdated over time. ViroPharma does not assume any responsibility for updating any forward-looking statements. These forward looking statements should not be relied upon as representing our assessments as of any date subsequent to the date of this press release.
SOURCE DuoCort Pharma
BACKGROUND: Bilateral adrenalectomy (BLA) is a treatment option to alleviate symptoms in patients with ectopic Cushing's syndrome (ECS) for whom surgical treatment of the responsible nonpituitary tumor is not possible. ECS patients have an increased risk for complications, because of high cortisol levels, poor clinical condition, and metabolic disturbances. This study aims to evaluate the safety and long-term efficacy of endoscopic BLA for ECS.
METHODS: From 1990 to present, 38 patients were diagnosed and treated for ECS in the Erasmus University Medical Center, a tertiary referral center. Twenty-four patients were treated with BLA (21 endoscopic, 3 open), 9 patients were treated medically, and 5 patients could be cured by complete resection of the adrenocorticotropic hormone (ACTH)-producing tumor. The medical records were retrospectively reviewed and entered into a database. For evaluation of the efficacy of BLA, preoperative biochemical and physical symptoms were assessed and compared with postoperative data.
RESULTS: Endoscopic BLA was successfully completed in 20 of the 21 patients; one required conversion to open BLA. Intraoperative complications occurred in two (10%) patients, and postoperative complications occurred in three (14%) patients. Median hospitalization was 9 (2-95) days, and median operating time was 246 (205-347) min. Hypercortisolism was resolved in all patients. Improvements of hypertension, body weight, Cushingoid appearance, impaired muscle strength, and ankle edema were achieved in 87, 90, 65, 61, and 78% of the patients, respectively. Resolution of diabetes, hypokalemia, and metabolic alkalosis was achieved in 33, 89, and 80%, respectively.
CONCLUSION: Endoscopic BLA is a safe and effective treatment for patients with ectopic Cushing's syndrome.
From http://www.docguide.com/endoscopic-bilateral-adrenalectomy-patients-ectopic-cushings-syndrome?tsid=5
Labels: ACTH, adrenalectomy, BLA, Cushing's, ectopic, endoscopic, hypercortisolism, surgery
MENLO PARK, CA, Nov 07, 2011 (MARKETWIRE via COMTEX) -- Corcept Therapeutics IncorporatedCORT -1.58% , a pharmaceutical company engaged in the discovery, development and commercialization of drugs for the treatment of severe metabolic and psychiatric disorders, today reported financial results for the quarter ended September 30, 2011, and updated its corporate progress.
"Following the acceptance by the U.S. Food and Drug Administration (FDA) of our New Drug Application (NDA) for the use of our lead product candidate, Korlym(TM), in Cushing's Syndrome," said Joseph Belanoff, M.D., Chief Executive Officer of Corcept, "we continue to focus our efforts on building our commercial capabilities to support the launch of Korlym, if Korlym is approved by the FDA, in order to allow us to provide an important treatment option to patients suffering from Cushing's Syndrome."
Corporate and Development Highlights
-- Received notification in October 2011 that the FDA had accepted our proposed brand name, Korlym (formerly referred to as CORLUX(R)), for our lead product candidate in the treatment of endogenous Cushing's Syndrome. -- Advanced our commercial launch preparations related to Korlym for the treatment of Cushing's Syndrome, including developing our internal infrastructure and engaging third-party vendors to provide market analytics and to support distribution and other logistical needs in the event Korlym is approved by the FDA. -- Received notification in October 2011 that the European Commission had granted Korlym Orphan Designation for the treatment of endogenous Cushing's Syndrome (hypercortisolism) in the European Union (EU). Benefits of Orphan Drug Designation in the EU are similar to those in the U.S., but include ten years of marketing exclusivity in all 27 member states, free scientific advice during drug development, access to a centralized review process and a reduction or complete waiver of fees levied by the European Medicines Agency. -- Enrolled additional patients in our double-blind placebo controlled Phase 3 trial of Korlym for the treatment of the psychotic features of psychotic depression. -- Continued the clinical portion of our Phase 1b/2a multi-dose safety and proof of concept studies of CORT 108297, one of our selective GR-II antagonists. -- Identified additional compounds from among our proprietary series of selective GR-II antagonists to advance toward an Investigational New Drug submission.
Third Quarter Financial Results
For the third quarter of 2011, Corcept reported a net loss of $6.4 million, or $0.08 per share, compared to a net loss of $7.1 million, or $0.10 per share, for the third quarter of 2010.
In the third quarter of 2011, research and development expenses decreased to $3.2 million from $5.2 million in the third quarter of 2010. This decrease in research and development expenses was due primarily to decreases in clinical trial costs related to drug-drug interaction and other NDA-supportive studies with Korlym, which were substantially completed in late 2010, and decreases in the clinical trial costs related to the Phase 1b/2a studies with CORT 108297. These decreases were partially offset by increased costs associated with the prosecution of our NDA for Korlym for the treatment of Cushing's Syndrome. General and administrative expenses increased to $3.2 million for the third quarter of 2011 from $1.9 million for the same period in 2010 due primarily to additional expenditures on commercialization activities for the potential launch of Korlym for Cushing's Syndrome.
Our cash balance as of September 30, 2011 was $45.9 million, up from $24.6 million at December 31, 2010. "We anticipate that our current cash balance is sufficient to fund the company through the end of 2012," said Charles Robb, the company's Chief Financial Officer.
Anticipated Activities for the Remainder of 2011
We continue to concentrate our efforts on advancing Korlym toward approval and commercialization for the treatment of Cushing's Syndrome. We also continue our efforts to be prepared to respond in a timely fashion to any questions posed by the FDA during the course of their review of our NDA.
"We are focused intently on developing the commercial and logistical capabilities we will need to make Korlym available to patients suffering from Cushing's Syndrome, should the FDA approve our drug for this indication," added Dr. Belanoff. "Korlym is the first step in unlocking the value of our scientific platform. The regulation of cortisol is a critical biological function; its dysregulation is equally critical in many important disease states. Our own research and research from increasing numbers of academic investigators point to the potential importance of cortisol antagonism in a wide variety of diseases. We believe our expanding library of selective cortisol antagonists may help address these unmet medical needs."
About Cushing's Syndrome
Endogenous Cushing's Syndrome is caused by prolonged exposure of the body's tissues to high levels of the hormone cortisol and is generated by tumors that produce cortisol or ACTH. Cushing's Syndrome is an orphan indication which most commonly affects adults aged 20 to 50. An estimated 10 to 15 of every one million people are newly diagnosed with this syndrome each year, resulting in over 3,000 new patients in the United States. An estimated 20,000 patients in the United States have Cushing's Syndrome. Symptoms vary, but most people have one or more of the following manifestations: high blood sugar, diabetes, high blood pressure, upper body obesity, rounded face, increased fat around the neck, thinning arms and legs, severe fatigue and weak muscles. Irritability, anxiety, cognitive disturbances and depression are also common. Cushing's Syndrome can affect every organ system in the body and can be lethal if not treated effectively.
About Psychotic Depression
Psychotic depression is a serious psychiatric disorder that affects approximately three million people annually in the United States. It is more prevalent than either schizophrenia or bipolar I disorder. The disorder is characterized by severe depression accompanied by delusions, hallucinations or both. People with psychotic depression are approximately 70 times more likely to commit suicide than the general population and often require lengthy and expensive hospital stays. There is no FDA-approved treatment for psychotic depression.
About Weight Gain Caused by Antipsychotic Medications
The group of medications known as second-generation antipsychotics, including olanzapine (Zyprexa), risperidone (Risperdal), quetiapine (Seroquel) and clozapine (Clozaril), are widely used to treat schizophrenia and bipolar disorder. All medications in this group are associated with treatment emergent weight gain of varying degrees and also carry warning labels relating to treatment emergent hyperglycemia and diabetes mellitus. There is no FDA-approved treatment for the weight gain associated with the use of antipsychotic medications.
About Korlym
Corcept's first-generation compound, Korlym, also known as mifepristone, directly blocks the cortisol (GR-II) receptor and the progesterone (PR) receptor. Intellectual property protection is in place to protect important methods of use for Korlym. Corcept retains worldwide rights to its intellectual property related to Korlym.
About CORT 108297
CORT 108297 is a potent, selective antagonist of the cortisol (GR-II) receptor that we have discovered and for which Corcept owns worldwide intellectual property rights. In in vitro binding affinity and functional assays this compound has no affinity for the progesterone (PR), estrogen (ER), androgen (AR) or mineralocorticoid (GR-I) receptors.
About Corcept Therapeutics Incorporated
Corcept is a pharmaceutical company engaged in the discovery, development and commercialization of drugs for the treatment of severe metabolic and psychiatric disorders. The company has completed its Phase 3 study of Korlym for the treatment of Cushing's Syndrome, and has an ongoing Phase 3 study of Korlym for the treatment of the psychotic features of psychotic depression. Corcept also has a Phase 2 program for CORT 108297, a selective GR-II antagonist that blocks the effects of cortisol but not progesterone. Corcept has developed an extensive intellectual property portfolio that covers the use of GR-II antagonists in the treatment of a wide variety of psychiatric and metabolic disorders, including the prevention of weight gain caused by the use of antipsychotic medication, as well as composition of matter patents for our selective GR-II antagonists.
Statements made in this news release, other than statements of historical fact, are forward-looking statements, including, for example, statements relating to the potential benefit of Korlym for patients diagnosed with Cushing's Syndrome, Corcept's clinical development and research programs, the outcome of the FDA's review of our NDA filing, our estimates for our capital requirements and needs for additional financing, the introduction of Korlym and future product candidates, including CORT 108297, the ability to create value from Korlym or other future product candidates or our scientific platform and our commercialization plans. Forward-looking statements are subject to a number of known and unknown risks and uncertainties that might cause actual results to differ materially from those expressed or implied by such statements. For example, there can be no assurances with respect to the cost, rate of spending, completion or success of clinical trials; financial projections may not be accurate; there can be no assurances that Corcept will pursue further activities with respect to the development of Korlym, CORT 108297, or any of its other selective GR-II antagonists. These and other risk factors are set forth in the Company's SEC filings, all of which are available from our website ( www.corcept.com ) or from the SEC's website ( www.sec.gov ). We disclaim any intention or duty to update any forward-looking statement made in this news release.
CORCEPT THERAPEUTICS INCORPORATED CONDENSED BALANCE SHEETS (in thousands) September 30, December 31, 2011 2010 -------------- -------------- (Unaudited) (Note) ASSETS: Current assets: Cash and cash equivalents $ 45,909 $ 24,578 Other current assets 427 418 -------------- -------------- Total current assets 46,336 24,996 Other assets 43 108 -------------- -------------- Total assets $ 46,379 $ 25,104 ============== ============== LIABILITIES AND STOCKHOLDERS' EQUITY: Current liabilities: Accounts payable $ 1,066 $ 817 Other current liabilities 1,647 3,043 -------------- -------------- Total current liabilities 2,713 3,860 Total stockholders' equity 43,666 21,244 -------------- -------------- Total liabilities and stockholders' equity $ 46,379 $ 25,104 ============== ============== Note: Derived from audited financial statements at that date. CORCEPT THERAPEUTICS INCORPORATED CONDENSED STATEMENTS OF OPERATIONS (in thousands, except per share amounts) (Unaudited) For the Three Months Ended For the Nine Months Ended September 30, September 30, -------------------------- -------------------------- 2011 2010 2011 2010 ------------ ------------ ------------ ------------ Operating expenses: Research and development* $ 3,228 $ 5,224 $ 14,355 $ 14,286 General and administrative* 3,209 1,881 8,049 5,327 ------------ ------------ ------------ ------------ Total operating expenses 6,437 7,105 22,404 19,613 ------------ ------------ ------------ ------------ Loss from operations (6,437) (7,105) (22,404) (19,613) Interest and other income, net 3 4 3 758 Other expense (1) (3) (17) (18) ------------ ------------ ------------ ------------ Net loss $ (6,435) $ (7,104) $ (22,418) $ (18,873) ============ ============ ============ ============ Basic and diluted net loss per share $ (0.08) $ (0.10) $ (0.27) $ (0.28) ============ ============ ============ ============ Shares used in computing basic and diluted net loss per share 84,188 72,045 83,000 66,982 ============ ============ ============ ============ *Includes non-cash stock-based compensation of the following: Research and development $ 110 $ 45 $ 432 $ 170 General and administrative 844 500 1,971 1,361 ------------ ------------ ------------ ------------ Total non-cash stock-based compensation $ 954 $ 545 $ 2,403 $ 1,531 ============ ============ ============ ============
CONTACT: Charles Robb Chief Financial Officer Corcept Therapeutics 650-688-8783 Email Contact www.corcept.com
SOURCE: Corcept Therapeutics
http://www2.marketwire.com/mw/emailprcntct?id=150008C85C40D638 http://www.corcept.com/
Labels: ACTH, Corlux, CORT 108297, cortisol, Cushing's, depression, FDA, Korlym, Mifepristone, progesterone, weight
Among the important early researchers in X-rays were Professor Ivan Pulyui, Sir William Crookes, Johann Wilhelm Hittorf, Eugen Goldstein, Heinrich Hertz, Philipp Lenard, Hermann von Helmholtz, Nikola Tesla, Thomas Edison, Charles Glover Barkla, Max von Laue, and Wilhelm Conrad Röntgen.

 Diagram of a water cooled X-ray tube. (simplified/outdated) In 1895, Thomas Edison investigated materials' ability to fluoresce when exposed to X-rays, and found that calcium tungstate was the most effective substance. Around March 1896, the fluoroscope he developed became the standard for medical X-ray examinations. Nevertheless, Edison dropped X-ray research around 1903 after the death of Clarence Madison Dally, one of his glassblowers. Dally had a habit of testing X-ray tubes on his hands, and acquired a cancer in them so tenacious that both arms were amputated in a futile attempt to save his life. "At the 1901 Pan-American Exposition in Buffalo, New York, an assassin shot President William McKinley twice at close range with a .32 caliber revolver." The first bullet was removed but the second remained lodged somewhere in his stomach. McKinley survived for some time and requested that Thomas Edison "rush an X-ray machine to Buffalo to find the stray bullet. It arrived but wasn't used . . . McKinley died of septic shock due to bacterial infection."[24]
Diagram of a water cooled X-ray tube. (simplified/outdated) In 1895, Thomas Edison investigated materials' ability to fluoresce when exposed to X-rays, and found that calcium tungstate was the most effective substance. Around March 1896, the fluoroscope he developed became the standard for medical X-ray examinations. Nevertheless, Edison dropped X-ray research around 1903 after the death of Clarence Madison Dally, one of his glassblowers. Dally had a habit of testing X-ray tubes on his hands, and acquired a cancer in them so tenacious that both arms were amputated in a futile attempt to save his life. "At the 1901 Pan-American Exposition in Buffalo, New York, an assassin shot President William McKinley twice at close range with a .32 caliber revolver." The first bullet was removed but the second remained lodged somewhere in his stomach. McKinley survived for some time and requested that Thomas Edison "rush an X-ray machine to Buffalo to find the stray bullet. It arrived but wasn't used . . . McKinley died of septic shock due to bacterial infection."[24] 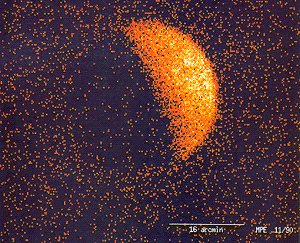

ROSAT image of X-ray fluorescence of, and occultation of the X-ray background by, the Moon. The use of X-rays for medical purposes (to develop into the field of radiation therapy) was pioneered by Major John Hall-Edwards in Birmingham, England. In 1908, he had to have his left arm amputated owing to the spread of X-ray dermatitis[1].
The X-ray microscope was invented in the 1950s. The Chandra X-ray Observatory, launched on July 23, 1999, has been allowing the exploration of the very violent processes in the universe which produce X-rays. Unlike visible light, which is a relatively stable view of the universe, the X-ray universe is unstable, it features stars being torn apart by black holes, galactic collisions, and novas, neutron stars that build up layers of plasma that then explode into space. An X-ray laser device was proposed as part of the Reagan Administration's Strategic Defense Initiative in the 1980s, but the first and only test of the device (a sort of laser "blaster", or death ray, powered by a thermonuclear explosion) gave inconclusive results. For technical and political reasons, the overall project (including the X-ray laser) was de-funded (though was later revived by the second Bush Administration as National Missile Defense using different technologies).Labels: Medical History, X-ray
Key points include: Advances and more frequent use of diagnostic radiology have led to the increased prevalence of endocrine incidentalomas; Pituitary, thyroid, and adrenal incidentalomas must be assessed for dysfunctional hormone secretion and malignant potential; Inpatient management of endocrine incidentalomas should include consultation of endocrine or surgical teams in cases of dysfunctional or malignant lesions; Post-discharge instructions shouldclearly delineate timelines for repeat imaging, laboratory testing, and subspecialist follow-up.
by Darlene Tad-y, MD, Section of Hospital Medicine, University of Colorado Denver

A 54-year-old man with a history of hypertension treated with hydrocholorothiazide and Type 2 diabetes mellitus is admitted with abdominal pain and found to have an incidental 2.1-cm left adrenal mass on CT scan of the abdomen. He denies symptoms of headache, palpitations, weight gain, or muscle weakness. His exam is significant for mildly elevated blood pressure. What is the best approach for evaluation and management of this incidental finding?
Incidentalomas are mass lesions that are inadvertently discovered during radiolographic diagnostic testing or treatment for other clinical conditions that are unrelated to the incidental mass. In recent decades, improvements in radiographic diagnostic techniques and sensitivity have led to increasing discovery of incidental lesions that are often in the absence of clinical signs or symptoms.1 Three commonly discovered lesions by hospitalists are pituitary, thyroid, and adrenal incidentalomas.2 The concerns associated with these findings relate to the potential for dysfunctional hormone secretion or malignancy.
Patients found with pituitary incidentalomas can be susceptible to several types of adverse outcomes: hormonal hypersecretion, hypopituitarism, neurologic morbidity due to tumor size, and malignancy in rare cases. Thyroid incidentalomas are impalpable nodules discovered in the setting of ultrasound or cross-sectional neck scans, such as positron emission tomography (PET) scans. Discovery of a thyroid incidentaloma raises concern for thyroid malignancy.3 The increased use of abdominal ultrasound, CT scans, and MRI has fueled the growing incidence of adrenal incidentalomas (AIs).
The discovery of an endocrine incidentaloma in the inpatient setting warrants a systematic approach that includes both diagnostic and potentially therapeutic management. A hospitalist should consider an approach that includes (see Table 1):
Pituitary incidentalomas. The prevalence of pituitary incidentalomas found by CT ranges from 3.7% to 20%, while the prevalence found by MRI approximates 10%. Autopsy studies have revealed a prevalence ranging from 1.5% to 26.7% for adenomas less than 10 mm, considered to be microadenomas. Broad categories of etiologies should be considered: pituitary adenoma, nonpituitary tumors, vascular lesions, infiltrative disorders, and others (see Table 2). The majority of pituitary adenomas secrete prolactin (30% to 40%) or are nonsecreting (30% to 40%). Adenomas secreting adrenocorticotropin hormone (ACTH, 2% to 10%), growth hormone (GH, 2% to 10%), thyroid-stimulating hormone (TSH, <1%), follicle-stimulating hormone (FSH), and luteinizing hormone (LH) are much less common.2 Significant morbidity and premature mortality are associated with hyperprolactinemia, acromegaly (growth hormone excess), Cushing’s syndrome, and hyperthyroidism. Additionally, up to 41% of patients with macroadenomas were found to have varying degrees of hypopituitarism due to compression of the hypothalamus, the hypothalamic-pituitary stalk, or the pituitary itself.4
Pituitary adenoma:
Non-pituitary tumors:
Vascular lesions:
Infiltrative:
Other:
ACTH=adrenocorticotropin hormone, GH=growth hormone, TSH=thyroid-stimulating hormone (thyrotropin), LH=luteinizing hormone, FSH=follicle-stimulating hormone
Recently, the Endocrine Society released consensus recommendations to guide the evaluation and treatment of pituitary incidentalomas, which are included in the approach outlined below.5 A detailed history and physical examination should be obtained with specific inquiry as to signs and symptoms of hormonal excess and mass effect from the tumor. Examples of symptoms of hormone excess can include:
Symptoms related to the mass effect of the tumor include visual field defects and hypopituitarism related to the deficient hormone, including:
The size and location of the pituitary lesion must be assessed. Lesions greater than 10 mm are considered macroademonas, and their size will affect their management. If the lesion was initially identified by CT scan, an MRI is recommended to better evaluate it.5 If the MRI locates the incidentaloma abutting the optic nerve or chiasm, then the patient should undergo a formal visual field examination.
Indications for an inpatient surgical referral for treatment include: a lesion larger than 2 cm, evidence of mass effect such as visual field defects, neurologic compromise, opthalmoplegia, hypopituitarism, a tumor abutting the optic nerve or chiasm, pituitary apoplexy, and hypersecretion of hormones other than prolactin. Patients with prolactinomas warrant an inpatient endo-crinology consult and may need medical management with a dopamine agonist. Hormone replacement therapy can also be provided for patients with hypopituitarism.2,5
For patients who do not meet the criteria for inpatient surgical therapy, follow-up management must be arranged at the time of discharge. Clinical, laboratory assessment, and an MRI should be scheduled six months after the initial finding of the incidentaloma with the patient’s PCP or with an endocrinologist.5
Thyroid incidentalomas. The prevalence of thyroid nodules based on ultrasound studies ranges from 19% to 46%, with autopsy studies estimating an incidence of approximately 50%.2,6 Incidence of thyroid nodules also increases with age, as almost 60% of people over the age of 60 harbor a thyroid incidentaloma. The rate of malignancy in the general population has ranged between 8% and 24%; however, in the last decade, the rates have increased by 2.4 times as more sophisticated ultrasound techniques and liberal use of fine-needle aspiration (FNA) biopsies have detected subclinical disease.7,8
Etiologies for incidental thyroid nodules can be divided into benign and malignant causes. Benign etiologies include thyroid cyst (simple or complex), multinodular goiter, and Hashimoto’s thryoiditis, while malignant causes include papillary, medullary, follicular, Hurthle cell, and anaplastic carcinomas, thyroid lymphomas, and rare instances of metastatic cancers.2,3
Targeted history and physical examination helps to characterize the thyroid incidentaloma. Historical features, such as palpitations, weight loss, anxiety, new onset atrial fibrillation, or menstrual irregularities, coupled with tachycardia, tremors, proximal muscle weakness, and a palpable nodule aid in the diagnosis of hyperthyroidism. Findings such as a family history of thyroid cancer, symptoms of hoarseness or dysphagia, rapid growth of the nodule, environmental or history of head or neck irradiation along with physical findings of a hard, fixed nodule, or cervical lymphadenopathy increase the suspicion for malignancy.2,7
The functionality of the nodule can be assessed by checking TSH, free T3, and free T4 levels. Suppression of TSH (< 0.1 mU/L) with elevated levels of free T3 and T4 indicates nodule production of excess thyroid hormone and warrants thyroid scintography. Thyroid scintography will identify the nodule as “hot” (hyperfunctioning) or “cold” (nonfunctioning).2
Regardless of the radiographic modality that initially identified the thyroid incidentaloma, a dedicated thyroid high-resolution ultrasound should be ordered to assess the size, multiplicity (single or multinodular), location, and character (solid, cystic, or mixed).7
Recommendations for proceeding to FNA to evaluate for malignancy differ among subspecialty societies. Generally, nodules larger than 1 cm or nodules smaller than 1 cm with risk factors for malignancy should be referred for FNA.2,7
If diagnostic workup identifies a patient with hyperthyroidism due to an autonomously functional nodule or a nodule that may be at high risk for malignancy, it is appropriate to involve an endocrinologist and possibly a surgical subspecialist prior to discharge. Management of hyperthyroidism can include starting antithyroid agents (methimazole or propylthiouracil), radioactive iodine ablation, or referral for surgery.
Preparation for discharge of the patient whose incidentaloma is nonfunctional or does not appear to be malignant should include appointments to recheck thyroid hormone levels, including TSH as well as a thyroid ultrasound within one year of the initial discovery.
Adrenal incidentaloma. The prevalence of AIs found by CT of the abdomen ranges from 0.4% to 4%, while autopsy studies have found a prevalence of 1.4% to 9% with increasing prevalence with age.2,9,10 The majority of AIs are benign and nonfunctioning adenomas, in the absence of known malignancy. Other differential diagnoses include Cushing’s syndrome, pheochromocytoma, adrenocortical adenoma, aldosteronoma, and metastatic lesions.
Because functioning adrenal incidentalomas may be clinically silent, any patient found with an AI must undergo biochemical workup as part of their evaluation to assess for pheochromocytoma, Cushing’s syndrome, and if he or she has a history of hypertension or hyperaldosteronism (Conn’s syndrome). Table 3 outlines the approach for characterizing adrenal incidentalomas.2,11,12 An important point is that imaging studies are not useful in distinguishing a functioning versus nonfunctioning tumor but rather can help to discriminate malignant lesions.11
Inpatient surgical consult for resection is indicated if the patient is found to have pheochromocytoma, clinically apparent functioning adrenocortical adenoma, or a tumor size greater than 4 cm. Consultation with an endocrinologist is also recommended if biochemical tests are positive. If the diagnostic workup leads to suspicion for infection or metastatic disease, the patient should be referred for FNA.2,12
For patients whose lesions do not require surgical resection, repeat CT scan of the abdomen is recommended six months from the initial finding. Hospitalists should also arrange for the patient to repeat biochemical testing, including an overnight dexamethasone test.12,13
The patient underwent biochemical testing and was found to have normal levels of plasma-free metanephrines, a plasma aldosterone, plasma renin activity ratio less than 20, and a serum cortisol level of 7 mg/dL after his overnight dexamethasone suppression test. The 24-hour urine collection for free cortisol revealed elevated levels of cortisol in the urine, and the ACTH level was low.
Endocrinology and endocrine surgery teams were consulted, and recommended surgical resection. After surgical resection of his tumor, the patient was started on glucocorticoid replacement and was discharged with a follow-up appointment with endocrinology.
An inpatient approach to endocrine incidentalomas should include characterization of the clinical signs and symptoms, size, function, and malignant potential of the lesion. Based on this, inpatient surgical or medical management can be determined. Post-discharge management should include arrangements for surveillance testing and follow-up with appropriate subspecialists.
Dr. Tad-y is assistant professor of medicine and a hospitalist at the University of Colorado Denver.